An animal research program encompasses lots of different staff members, requirements, and details to ensure everything is done right. One aspect of this is training. In order to do animal research, lab members must have current training certifications for everything that will be involved, in order to ensure safe lab practices. And vivarium staff must be certified, too. Some common training certifications include:
- Vivarium orientation
- IACUC basics
- Lab safety
- Animal handling and husbandry
- Biological materials safety
- Hazardous waste management
- Radiation and laser safety
- Using personal protective equipment (PPE)
- Other equipment and sanitation training
These trainings not only keep staff safe, they also protect animal health, as well as protecting the program if an audit or inspection happens. Inspectors or auditors from the USDA, PHS, or other governmental agencies want to make sure staff members are trained for the work they do. Plus, the research program might also seek voluntary accreditation by the Association for Assessment and Accreditation of Laboratory Animal Care (AAALAC).
The challenge: keeping track of trainings
Keeping track of everyone’s qualifications is important not only for protocols and specific studies, but also for inspections and evaluations. When you have lots of staff members, this becomes a monumental task. Training is not a one-and-done thing, either. Continuing education, such as yearly seminars or triennial recertification, is often required in addition to one-time certifications.
It’s easier to manage training records with software, but many programs use paper files or a spreadsheet. The last two methods are really time consuming, since data entry and reporting have to be done manually. Managers (or the IACUC office or researchers, depending on your program) have to match the staff member’s training certification with the work they will be doing while keeping in mind when the certification will expire.
Sick days, new hires, and unexpected absences make it even more complicated. A well-meaning staff member may volunteer to fill in for a coworker but lack the necessary training. That may be an effective short-term solution, but it leaves the program vulnerable to audit findings for noncompliance. Nobody wants to risk noncompliance—it can result in loss of funding, whistleblower allegations, or being shut down altogether.
How Cayuse can help safeguard institutions
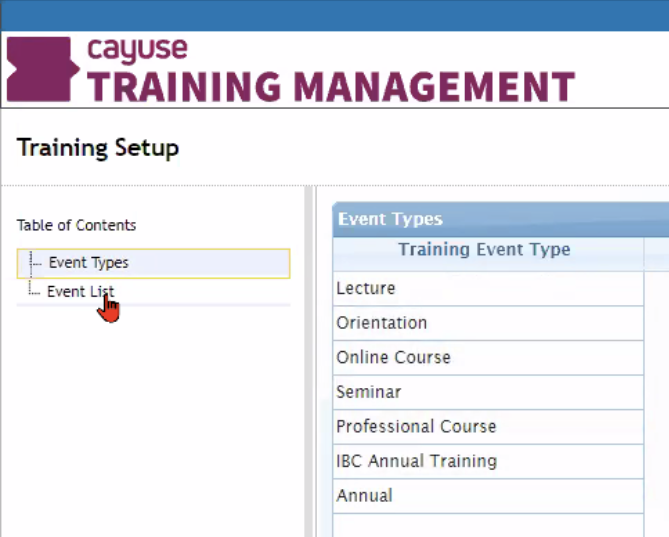
The best way to track staff certifications, improve compliance, and ensure everyone’s safety is to use Training Records, Animal Oversight, and Hazard Safety. They are intuitive solutions that help save time, improve safety, and lower the risk of noncompliance.
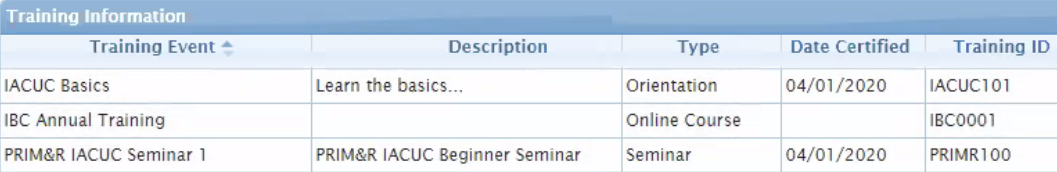
Here’s how it works. When researchers are creating protocols in Animal Oversight or Hazard Safety, they enter all the related study activities (for example, rabbit nail clipping and skin biopsy). Training Records seamlessly integrates with Animal Oversight, so the researcher can see which staff in the system have the necessary training certifications, when they were certified, and when the certification expires.
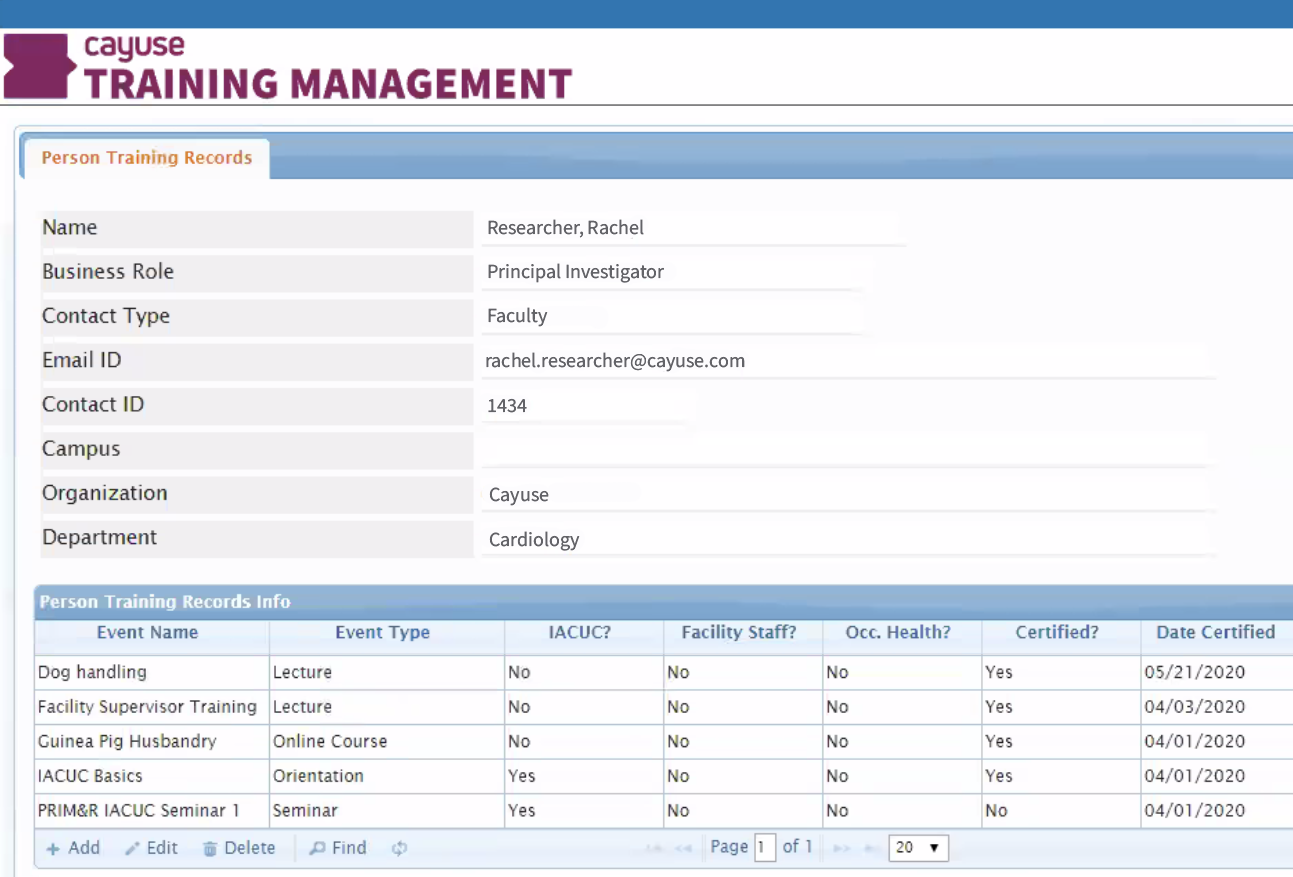
Researchers aren’t the only ones who benefit from Training Records. IACUC and IBC committees, training coordinators, and vivarium managers can also view personnel qualifications to ensure staff have completed the required procedure-specific training.
Training Records is also 100% paperless, so you can use it anywhere with an internet connection. Here are just a few things you can do with it:
- Set up training requirements by activities, species, procedures, and hazardous agents
- Track training at the personnel, species, and activity level
- Expedite protocol authorship and approval by automatically tracking training requirements
- Quickly and easily pull reports on-demand for reference and audit purposes
Ready to lower your noncompliance risk, make research safer, and save time for research staff? Learn more about Training Records, Animal Oversight, and Hazard Safety.


